scPharmaceuticals(SCPH)
Download report
Favorite
Financial
Marketed Drugs
Clinical Trials
Financial
Stock price
*Currency in USD

Company Revenue
Quarterly
Quarterly
Annual
Annual
Projections
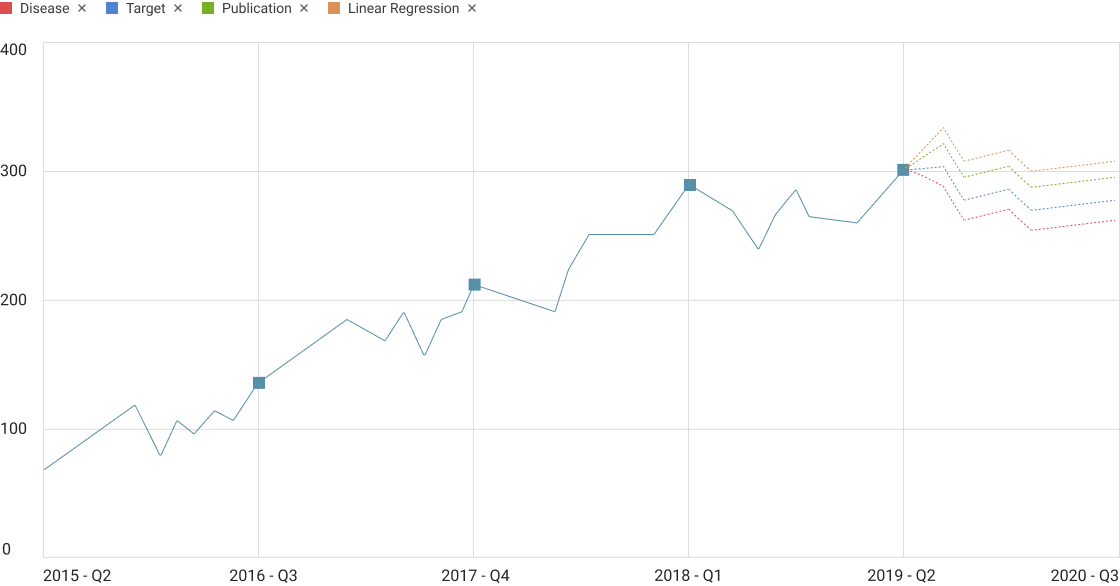
Mock data
Subscribe for the real data
Subscribe for the real data
Revenue by Drug

Marketed Drugs
Therapeutic Areas:
All
Classification:
All
Include Generics:
Trade Name | Drug Name | Approved | Patent Expires | Indication |
|---|---|---|---|---|
| Furoscix | Furosemide | 2022-10-07 | 2034 |
Clinical Trials
Therapeutic Areas:
All
Classification:
All
Approval:
All
Status:
All
Phases:
All
Drug Name | Phase 1 | Phase 2 | Phase 3 | Phase 4 | Indication |
|---|---|---|---|---|---|
© 2020-2025 Collaborative Drug Discovery Inc. (CDD) | Terms of Use